Official Listing of Recbio on HKEX, Vaccine Innovation and Iteration Accelerated by Novel Adjuvant Technology
 |
 |
 |
 |
Domestic vaccine innovation and iteration accelerated by novel adjuvant technology
Vaccines are praised as the “golden track” in the medical health field. According to the Frost & Sullivan report, from 2016 to 2020, the size of the global vaccine market has increased from US$27.5 million to US$39.9 billion at a CAGR of 9.7%, while the CAGR of global pharmaceutical market was only 3.02%. China has become the second largest vaccine market globally, with the market volume increased from RMB27.1 billion in 2016 to RMB75.3 billion in 2020 at a CAGR of 29.1%.
Regarding the insufficient domestic supply of and the unmet urgent demand for novel vaccines, it has become an important part of the Development Planning of the Pharmaceutical Industry in the “14th Five-Year Plan” in China (referred as the “Plan”) to promote enterprises to speed up the development of related products and to achieve product availability as soon as possible. Regarding the development of the vaccine industry, the “Plan” also states that research and development of novel vaccines (such as novel adjuvants for vaccines and mRNA vaccines) and building of industrialization capability shall be accelerated. It also emphasizes on the development of novel COVID-19 vaccines, herpes vaccines, polyvalent human papillomavirus (HPV) vaccines and other products.
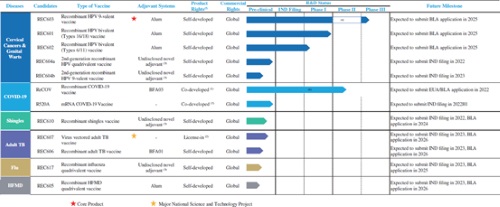
As an innovative novel vaccine company, Recbio has constructed the three core technology platforms consisting of novel adjuvants, protein engineering and immunological evaluation, forming synergy in antigen design and optimization, adjuvant development and production, identification of the best combination of antigen and adjuvants. The Company has built a comprehensive and high-valued vaccine portfolio consisting of 12 candidates, covering cervical cancer, COVID-19, adult tuberculosis, shingles, HFMD, influenza etc. It covers five of the ten diseases with the greatest burden under the 2019 Global Burden of Diseases issued by the World Health Organization and disease areas of the three of the top five globally bestselling vaccine products in 2020.
Novel adjuvant for vaccines is the “bottleneck” technology of innovative vaccines. Through years of technical research, Recbio has successfully achieved independent control of all FDA-approved novel adjuvants, and is one of the few companies in the world capable of independent development of novel adjuvants. It has also successfully applied in various innovative vaccines such as COVID-19 vaccines, HPV vaccines and shingles vaccines.
A moat constructed with a rich product portfolio
HPV vaccines are one of the most commercially valuable vaccines in the world. It is expected that there will be 234 million females in China aged 9-45 unvaccinated for HPV in 2025 assuming 3 doses per person, requiring additional 702 million doses. Currently, Recbio has formed a full coverage of bivalent, quadrivalent and 9-valent vaccines and novel adjuvant upgrades in the field of HPV vaccines. The core product REC603, a recombinant HPV 9-valent vaccine is currently under phase III clinical trial, expected to become the first approved domestic HPV 9-valent vaccine. Regarding the global immunization program market, REC601, a bivalent vaccine has the potential to become one of the most popular vaccine options. At the same time, the Company is also in the process of developing second-generation HPV vaccines, namely REC604a (a quadrivalent vaccine) and REC604b (a 9-valent vaccine). Both vaccines have adopted our self-developed novel adjuvants and are expected to achieve two-shot regimens in the future. The construction of the first phase of the HPV manufacturing facility of Recbio is expected to be completed by the end of this year. By then, it will realize the designed capacity of five million doses of HPV 9-valent vaccines or 30 million doses of HPV bivalent vaccines per year. As the first cancer preventive vaccine in the world, HPV vaccines have been in shortage for a long time. With the gradual release of domestic HPV vaccines in the future and the gradual release of production capacity, domestic substitution will be accelerated.
Protein engineering, novel adjuvant and other technology are comprehensively used in ReCOV, the COVID-19 vaccine of Recbio, to thoroughly optimize the vaccine, allowing ReCOV to possess a series of comprehensive advantages, including excellent safety, strong immunogenicity, strong cross-protection effectiveness with Omicron and Delta mutant strains, easy-to-scale-up production, low production cost, high preparation stability, storage and transportation in room temperature etc. The international phase II/III clinical trials of such vaccine are currently in progress. EUA/BLA applications are estimated to be submitted in 2022. On the side of production supply, the Company has completed the construction of our GMP-standard manufacturing facility for ReCOV and obtained the drug production license in November 2021. The manufacturing facility has a total GFA of approximately 17,000 sq.m., has the potential to support an annual manufacturing capacity of 300 million doses of ReCOV and can be used to manufacture recombinant shingles vaccines.
In the field of shingles vaccines, shingles virus is dormant in almost all adults over 50 of age in China, affecting approximately 2.5 million adults in China every year. However, Shingrix is currently the only shingles vaccine approved in China. The novel adjuvant which benchmarking AS01 is adopted in REC610 of the Company, significantly improving vaccine immunogenicity, hoping to achieve domestic substitution quickly.
Recogen, our subsidiary has first achieved technology breakthrough in lyophilized mRNA vaccines. It has applied the self-developed freeze-drying technology to achieve preparation stability at 4 degrees Celsius and 25 degrees Celsius, allowing storage and transportation under general cold-chain conditions, effectively solving the current defect in availability of mRNA vaccines.
“We are glad that the Hong Kong stock IPO has been highly recognized by the capital market. The successful listing of Recbio is expected to bring more confidence to the Hong Kong 18A stock market. Thanks to all the investors for their strong support, we were able to successfully go public as originally planned in such extremely depressed environment of the Hong Kong capital market!” Regarding the listing of Recbio in Hong Kong, Dr. Liu Yong, the Founder, Chairman of the Board, and General Manager of Recbio stated that, “Against the backdrop of a huge global demand for innovative vaccines in this era, through continuous innovation and international cooperation, we will accelerate the commercialization of products in the global market, promote innovation in the entire value chain of the vaccine industry from R&D to commercialization, and create greater value for shareholders and investors. Today is the beginning of a brand-new journey for Recbio. I hope that all investors will continue to support our Company. Let’s hope we will continue to move forward hand in hand, through the sweltering summer and winter, and create new glory together!”
About Recbio
Founded in 2012, Recbio is an innovative vaccine company with independent research and development technology as its core driving force. It is committed to building a full value chain of innovative vaccines, covering research and development, production and commercialization. The Company has always adhered to the mission of “creating first-class vaccines and protecting human health”. With a strong research and development engine comprising of novel adjuvants, protein engineering, immunological evaluation and other core technology platforms, strategically focusing on disease areas with significant burden such as cervical cancers COVID-19, shingles, adult TB, HFMD and flu etc., the Company has established a high-value product pipeline consisting of 12 vaccine candidates with independent intellectual property rights, in order to meet the under-fulfilled public health needs. For more information, please visit https://www.recbio.cn/.
Copyright 2022 ACN Newswire. All rights reserved. (via SEAPRWire)